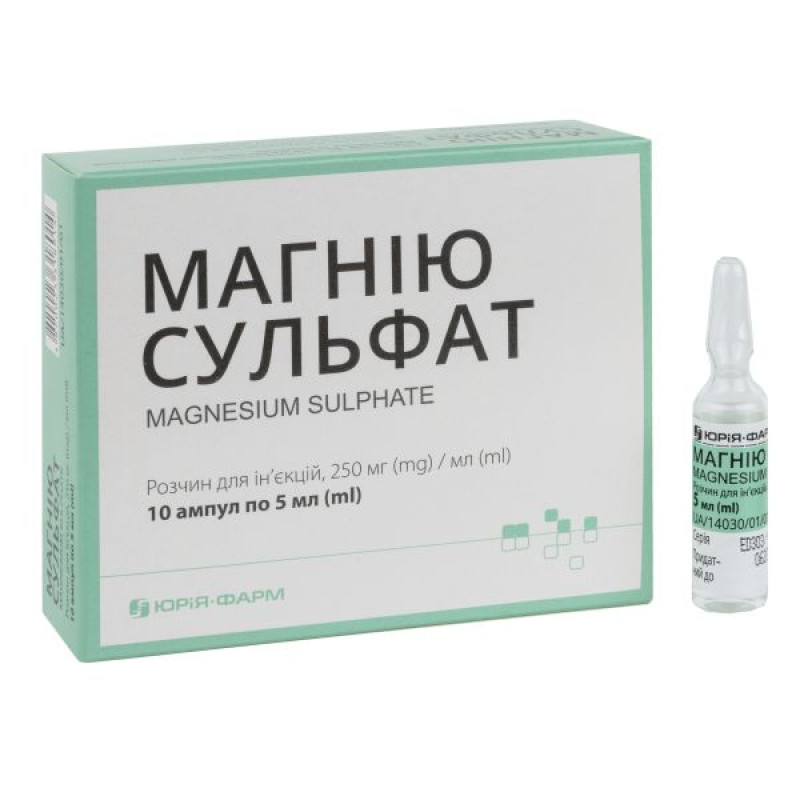
Magnesium sulfate solution for injection 25% ampoule 5 ml No. 10
Instructions Magnesium sulfate solution for injection 25% ampoule 5 ml No. 10
Composition
active ingredient: magnesium sulfate heptahydrate;
1 ml of solution contains 250 mg of magnesium sulfate heptahydrate;
excipient: sodium hydroxide, diluted hydrochloric acid, water for injections.
Dosage form
Solution for injection.
Main physicochemical properties: clear colorless liquid.
Pharmacotherapeutic group
Blood substitutes and infusion solutions. Electrolyte solution. ATX code B05X A05.
Pharmacological properties
Pharmacodynamics
Magnesium is a physiological antagonist of calcium, a metabolic cofactor of most metabolic reactions, including those associated with the synthesis and release of energy, reduces the secretion of catecholamines, regulates the functioning of Na+–K+ ATPase, neurochemical transmission, muscle excitability, reduces the content of acetylcholine in the central nervous system, peripheral nervous system, as a result of which it has a sedative, analgesic, anticonvulsant, spasmolytic, choleretic and tocolytic effect. The drug dilates coronary and peripheral arteries, reduces blood pressure, afterload on the heart, inhibits the development of reperfusion injury to the myocardium. Reduces the frequency of ventricular and supraventricular arrhythmias, slows down conduction in the sinus and atrioventricular nodes.
The antiplatelet properties of magnesium are associated with a decrease in the synthesis of thromboxane A2, lipoxygenase derivatives (12-HETE), stimulation of the synthesis of prostacyclin and high-density lipoproteins. At higher doses, magnesium can cause negative isotropic and muscle relaxant effects.
Pharmacokinetics
When administered parenterally, magnesium quickly enters organs and tissues, penetrates the blood-brain barrier, the placenta, and in high concentrations penetrates into breast milk. The drug is excreted mainly in the urine.
Systemic effects develop within 1 minute after intravenous administration and 1 hour after intramuscular administration. The duration of action of magnesium with intravenous administration is 30 minutes, with intramuscular administration – 3-4 hours.
Indication
Hypertensive crisis, ventricular arrhythmias (pirouette tachycardia); convulsive syndrome; eclampsia, hypomagnesemia, increased need for magnesium. In the complex therapy of premature birth, angina pectoris, poisoning with heavy metal salts, tetraethyl lead, soluble barium salts (antidote).
Contraindication
Increased individual sensitivity to the components of the drug; arterial hypotension; severe bradycardia (heart rate less than 55 beats per minute); atrioventricular block; conditions caused by calcium deficiency and depression of the respiratory center; cachexia; impaired renal function; severe hepatic or renal failure; myasthenia gravis; malignant neoplasms.
Interaction with other medicinal products and other types of interactions
Calcium ions have an antagonistic effect on magnesium ions, which leads to a decrease in the pharmacological effects of magnesium sulfate when used simultaneously. The drug enhances the effect of drugs that depress the central nervous system (narcotics, analgesics). With the simultaneous use of muscle relaxants and nifedipine, neuromuscular blockade is enhanced. Simultaneous use with calcium channel blockers, such as nifedipine, can lead to a violation of calcium balance and impaired muscle function.
Barbiturates, narcotic analgesics and antihypertensives increase the likelihood of respiratory depression. Cardiac glycosides increase the risk of conduction disturbances and atrioventricular block.
The effect of antithrombotic agents, vitamin K antagonists, isoniazid, and nonselective inhibitors of neuronal monoamine reuptake is reduced.
Mexiletine elimination may be slowed, which may require dose adjustment.
Propafenone: the effect of both drugs is enhanced and the risk of toxic effects increases.
Disrupts the absorption of tetracycline antibiotics, intestinal obstruction is possible, and weakens the effect of streptomycin and tobramycin.
Application features
When using the drug, it should be taken into account that increased urinary magnesium excretion occurs with an increase in extracellular fluid, dilation of renal vessels, hypercalcemia, increased urinary sodium excretion, when prescribing osmotic diuretics (urea, mannitol, glucose), "loop" diuretics (furosemide, ethacrynic acid, thiazides), when taking cardiac glycosides, calcitonin, thyroidin, with prolonged administration of deoxycorticosterone acetate (more than 3-4 days). Slowing down the excretion of magnesium is observed with the administration of parathyroid hormone. In renal failure, magnesium excretion slows down, and with repeated administrations its cumulation may occur. Therefore, for elderly patients and patients with severe renal impairment, the dose of the drug should not exceed 20 g of magnesium sulfate (81 mmol Mg2+) within 48 hours, and in patients with oliguria or severe renal impairment, magnesium sulfate should not be administered intravenously rapidly. Urinary tract infections accelerate the precipitation of ammonium-magnesium phosphates, and magnesium therapy is temporarily not recommended. In case of impaired magnesium excretion after parenteral administration of magnesium sulfate, hypermagnesemia is possible.
Use with caution in myasthenia gravis and respiratory diseases. With prolonged use of the drug, monitoring of the cardiovascular system, tendon reflexes, renal function and respiratory rate is recommended.
Intravenous administration of magnesium sulfate should be carried out slowly: if the rate of administration is too high, hypermagnesemia may occur (symptoms include nausea, paresthesias, sedation, hypoventilation up to apnea, decreased deep tendon reflexes). Simultaneous parenteral administration of vitamin B6 and insulin increases the effectiveness of magnesium therapy.
If simultaneous intravenous administration of magnesium sulfate and calcium preparations is necessary, they should be administered into different veins, taking into account that the level of magnesium depends on the level of calcium in the body.
Use during pregnancy or breastfeeding
During pregnancy, magnesium sulfate should be used with extreme caution, taking into account the concentration of magnesium in the blood in cases where the expected therapeutic effect outweighs the potential risk to the fetus. When providing analgesia during labor, the possibility of inhibiting the contractile ability of the uterine muscles should be taken into account, which requires the use of agents that stimulate labor.
If necessary, use of the drug should stop breastfeeding.
Ability to influence reaction speed when driving vehicles or other mechanisms
Patients should be warned about the need to be careful when working with potentially dangerous mechanisms or when driving vehicles, since the drug has a sedative effect.
Method of administration and doses
Administer intramuscularly, intravenously slowly or as an intravenous infusion. The frequency of administration and doses are individual, depending on the indications and therapeutic effect. When administered by infusion, the drug is diluted with 0.9% sodium chloride solution or 5% glucose. When administered intravenously, the rate of administration should usually not exceed 150 mg/min (0.6 ml/min), except for the treatment of arrhythmias and eclampsia in pregnant women.
Prepared infusion solutions must be used immediately after preparation (cannot be stored).
Hypomagnesemia. For moderately severe hypomagnesemia (0.5-0.7 mmol/L), adults should be administered 4 ml (1 g of magnesium sulfate) intramuscularly every 6 hours.
In severe hypomagnesemia (<0.5 mmol/l) with intramuscular administration, the total dose should be increased to 1 ml/kg (250 mg/kg) and administered in parts over 4 hours. In the form of intravenous infusion in severe hypomagnesemia, 20 ml of the drug (5 g of magnesium sulfate) should be added to 1 l of 0.9% sodium chloride solution or 5% glucose and administered over at least 3 hours.
The maximum daily dose for intravenous administration is 72 ml (18 g). If necessary, the infusion should be repeated within several days.
Arterial hypertension. In case of arterial hypertension of stage I-II, administer 5-10-20 ml intramuscularly daily. The course of treatment is 15-20 injections, while along with a decrease in blood pressure, a decrease in the severity of angina pectoris is possible.
Hypertensive crisis. Administer 10-20 ml intramuscularly or intravenously slowly.
Cardiac arrhythmias. To stop arrhythmias, inject 4-8 ml (1-2 g of magnesium sulfate) intravenously over 5-10 minutes, if necessary, repeat the injection (total injection - up to 4 g of magnesium sulfate). It is possible to administer initially in a loading dose of 8 ml over at least 5 minutes, followed by infusion of 20 ml of the drug diluted with 0.9% sodium chloride solution or 5% glucose over at least 6 hours, or initially 8 ml over at least 30 minutes, followed by infusion over at least 12 hours.
Ischemic stroke. 10-20 ml intravenously for 5-7 days.
Convulsive syndrome. Adults should be given 5-10-20 ml intramuscularly. Children should be given intramuscularly at the rate of 0.08-0.16 ml/kg of body weight (20-40 mg/kg).
In case of preeclampsia or eclampsia, administer intramuscularly or intravenously. First, inject 10 ml intramuscularly into each buttock once or 16 ml (4 g of magnesium sulfate) intravenously over 3-4 minutes. Then continue to administer 16-20 ml (4-5 g) intramuscularly every 4 hours or intravenously drip 4-8 ml/hour (1-2 g/hour) with constant monitoring of tendon reflexes and respiratory function. Continue therapy until the attack stops. The maximum daily dose is 40 g of magnesium sulfate, in case of impaired renal function - 20 g/48 hours.
Labor pain relief. 5-10-20 ml intramuscularly, if necessary, combine magnesium sulfate with analgesics.
Urinary retention. For urinary retention and lead colic, administer 5-10 ml of the drug intramuscularly or 5-10 ml of a 25% solution of magnesium sulfate diluted 5 times intravenously (also administer as an enema).
As an antidote. In case of intoxication with mercury, arsenic, tetraethyl lead, intravenously administer 5-10 ml of a 25% solution of magnesium sulfate diluted 2.5-5 times. In case of poisoning with soluble barium salts, administer 4-8 ml intravenously or rinse the stomach with a 1% solution of magnesium sulfate.
Newborns. In case of intracranial hypertension and severe asphyxia in newborns, administer intramuscularly, starting with a dose of 0.2 ml/kg of body weight per day, increasing the dose on the 3rd-4th day to 0.8 ml/kg of body weight per day for 3-8 days in complex therapy. To eliminate magnesium deficiency in newborns, prescribe 0.5-0.8 ml/kg 1 time per day for 5-8 days.
Children
The drug can be used in pediatric practice.
Overdose
Symptoms: signs of hypermagnesemia in order of increasing serum magnesium concentration:
- decreased deep tendon reflexes (2-3.5 mmol/l);
- prolongation of the PQ interval and expansion of the QRS complex on the ECG (2.5-5 mmol/l);
- loss of deep tendon reflexes (4-5 mmol/l);
- depression of the respiratory center (5-6.5 mmol/l);
- cardiac conduction disorders (7.5 mmol/l);
- cardiac arrest (12.5 mmol/l).
In addition, hyperhidrosis, anxiety, inhibition, polyuria, uterine atony.
Treatment: specific antidote is calcium preparations (calcium chloride or gluconate), which should be administered intravenously slowly. In case of moderate hypermagnesemia, furosemide may be prescribed. Respiratory depression should be eliminated by intravenous administration of 5-10 ml of 10% calcium chloride solution, oxygen inhalation, artificial ventilation of the lungs. In severe cases, peritoneal dialysis or hemodialysis is indicated. The appointment of symptomatic agents that correct the functions of the cardiovascular and central nervous systems.
Adverse reactions
Cardiovascular system: hypotension, bradycardia, palpitations, conduction disturbances, hot flashes/feelings of heat, prolongation of the PQ interval and widening of the QRS complex on the ECG, arrhythmia, coma, cardiac arrest.
Respiratory system: shortness of breath, respiratory depression.
Nervous system: headache, dizziness, general weakness, drowsiness, confusion, loss of consciousness, depressed mood, decreased tendon reflexes, diplopia, anxiety, speech disorders, tremor and numbness of the extremities.
Musculoskeletal system: muscle weakness.
From the digestive tract: nausea, vomiting.
Allergic reactions: anaphylactic shock, angioedema, hyperthermic syndrome, chills.
Skin: hyperemia, itching, rash, urticaria, increased sweating.
From the urinary system: polyuria.
From the reproductive system and mammary glands: uterine atony.
Metabolic disorders: hypocalcemia, hypophosphatemia, hyperosmolar dehydration.
Injection site disorders: hyperemia, edema, pain.
Expiration date
. 2 years.
Storage conditions
. Store at a temperature not exceeding 25 ºС in the original packaging. Do not freeze. Keep out of the reach of children.
Incompatibility
Pharmaceutically incompatible (precipitate is formed) with calcium preparations, ethanol (in high concentrations), carbonates, bicarbonates and phosphates of alkali metals, salts of arsenic acid, barium, strontium, clindamycin phosphate, hydrocortisone sodium succinate, polymyxin B sulfate, procaine hydrochloride, salicylates and tartrates. At Mg2+ concentrations of more than 10 mmol/ml in mixtures for total parenteral nutrition, stratification of fat emulsions is possible.
Packaging
5 ml or 10 ml in an ampoule; 5 ampoules in a contour blister pack; 2 contour blister packs in a cardboard pack.
Vacation category
According to the recipe.
Producer
LLC "Yuria-Pharm".
Location of the manufacturer and its business address
Ukraine, 18030, Cherkasy, Verbovetskogo St., 108. Tel. (044) 281-01-01
There are no reviews for this product.
There are no reviews for this product, be the first to leave your review.
No questions about this product, be the first and ask your question.